From Whey to Tagatose#
About the project#
Duration: 20–50 hours (including typesetting final report in LaTeX)
Prerequisites: Differential equations (incl. separable ODEs), Lambert W function, basic calculus (derivatives/integration), basic Python programming
Python packages: NumPy, matplotlib, sympy, scipy
Learning objectives: Model enzyme kinetics with Michaelis–Menten theory, analyze (non)linear ODE models with initial conditions, compare inhibited vs. non-inhibited kinetics, include time/temperature-dependent rate parameters, and optimize process conditions under constraints
1 Introduction#
Important
This project was created by Per Karlsson and Kim Knudsen, DTU Mathematics, in collaboration with Jørgen Risum, DTU Food.
In cheese production, a large amount of whey is produced, which contains protein and lactose. Immediately, whey constitutes a waste problem. Although whey is readily degradable in a biological wastewater treatment plant, establishing and operating such a plant is very expensive. A better option for “disposal” is based on processing whey into products of higher value. Whey proteins have properties that make them useful as ingredients in the food industry. They can be concentrated from whey by ultrafiltration. The remainder from the process is a solution of lactose (and minerals). Lactose has no particular value, since both sweetness and solubility are very limited.
The disaccharide lactose can be hydrolyzed into the monosaccharides glucose and galactose. Both are much sweeter than lactose and at the same time much more soluble. The increased sweetness can be utilized directly by evaporating the liquid into a syrup that is used in food products.
Alternatively, galactose can be converted to tagatose, which is an interesting product. Tagatose is a sugar with very high sweetness, which is difficult to absorb from the intestine, and which does not clump. Therefore, the product is interesting as a low-calorie sweetener.
Optionally read more about industrial production and use of tagatose on various websites.
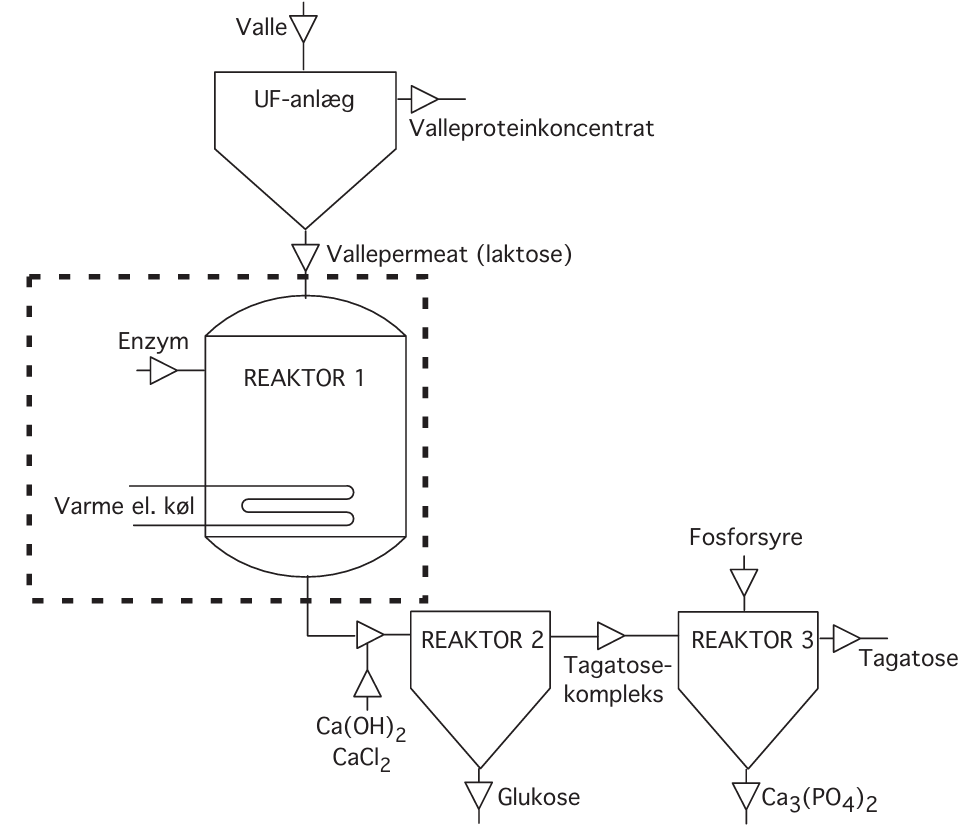
*Figure 1: Process sketch – the project assignment concerns what happens in reactor 1. Danish terms in the figure translate as follows: valle (whey), UF-anlæg (UF plant/Ultrafiltration unit), Valleproteinkoncentrat (whey protein concentrate), vallepermat (laktose) (whey permeate (lactose)), enzym (enzyme), varme e. køl (heating or cooling), reaktor (reactor), glukose (glucose), tagatose-kompleks (tagatose complex), and fosforsyre (phosphoric acid).
The process can briefly be described by the following subprocesses:
Cheesemaking:
Cheese milk ⇒ Cheese + Whey
Ultrafiltration:
Whey ⇒ Retentate (with protein) + Permeate (with lactose)
Drying:
Retentate ⇒ Whey protein concentrate
Hydrolysis with β-galactosidase:
Lactose ⇒ Glucose + Galactose
Isomerization at pH 12.5 (+ Ca(OH)₂ + CaCl₂):
⇒ Precipitate with tagatose + glucose solution
Dissolution of precipitate at pH 6.5:
⇒ Tagatose solution
The overall process is illustrated in Figure 1. The result is thus marketable products and a more limited waste problem mainly consisting of salt solutions. The basis for the process is that lactose is hydrolyzed to glucose and galactose. It is this part of the process that we will investigate in this project.
2 Enzyme kinetics#
For the hydrolysis, the enzyme β-galactosidase (EC. 3.1.2.23) is used, which catalyzes the process:
Lactose + H₂O ⇒ Glucose + Galactose
The equilibrium of the reaction is very strongly shifted to the right, since
is indeed very large.
The reaction rate can be described by the classical Michaelis–Menten expression
where
Symbol |
Quantity |
Unit |
|---|---|---|
\(v(t)\) |
Reaction rate |
M/min |
\(v_{\max}\) |
Maximum reaction rate |
M/min |
\(c(t)\) |
Substrate concentration |
M |
and \(K_m\) is a constant that describes affinity between substrate and enzyme. In the rest of the project assignment we assume that \(K_m = 6\ \text{mM}\). The maximum reaction rate, \(v_{\max}\), depends e.g. on the amount and strength of the added enzyme, the reaction time and temperature. However, we will initially assume that \(v_{\max}\) is constant.
The reaction rate is seen to depend on the substrate concentration, \(c=c(t)\).
1. Set up an expression that describes \(v\) when \(c \ll K_m\) and when \(c \gg K_m\).
2. Draw the graph (use Sympy) for \(v\) as a function of \(c\) (e.g. with \(v_{\max}=100\ \mu\text{M/min}\)).
The initial concentration of substrate \(c_0\) is known, and together with equation (2) it determines \(c(t)\) for later times.
3. Argue that equation (2) with an initial condition \(c(0)=c_0>0\) has a uniquely determined solution. Show that \(c(t)>0\) for \(t>0\).
4. Argue from the differential equation (2) that \(c(t)\) has an inverse function \(t(c)\). Derive a differential equation for \(t(c)\) and state an initial condition.
5. Using the method of separation of variables, set up an expression for the function \(t(c)\), which determines how long it takes to reach a given concentration \(c\).
6. Use the result in problem 5 as well as the problems about the Lambert W function to determine \(c(t)\) when the initial condition is \(c(0)=c_0\).
7. Plot the two expressions for \(c_0=0.15\ \text{M}\), \(K_m=6\ \text{mM}\) and \(v_{\max}=100\ \mu\text{M/min}\).
8. What happens to the concentration when \(t\to\infty\)? Is it consistent with the statement about the constant \(K\) in (1)?
Two expressions have now been set up for the integrated Michaelis–Menten equation. In the literature one often finds the relation between concentration and time expressed by
9. Do the two found expressions match this?
3 Inhibition#
Enzymes catalyze conversion of substances, and the products often resemble the substrate. Therefore, one can imagine that a competition arises between substrate and product for the site on the enzyme where molecules bind. When this is the case, the reaction is said to be inhibited. Since it is competition, and since it is the product that inhibits, it is called competitive product inhibition. The phenomenon is of course unfortunate when it is the product that is the interesting one, as in the cleavage of lactose. Here it holds that galactose acts as a competitive inhibitor, whereas glucose apparently has no inhibiting effect.
The rate expression for a competitively product-inhibited reaction has the form
where \(K_I\) is a constant that describes the inhibition. In the rest of the project we set \(K_I=30\ \text{mM}\).
As for the process described by (2), an initial concentration \(c(0)=c_0\) determines the future concentration \(c(t)\). Furthermore, \(c(t)\) has an inverse function \(t(c)\), which gives the time it takes to reach concentration \(c\).
10. Analyze the differential equation (4) by answering Problems 2–8 with equation (2) replaced by equation (4). Use \(c_0=0.15\ \text{M}\). Compare the results for the inhibited process with the results for the non-inhibited process.
The integrated expression from the literature is:
11. Does this expression match the found integrated expressions?
With the expressions now developed we can provide a preliminary solution to the task: In order to be able to carry out a sizing, experiments are performed on a larger scale to find out how long it will take to reach a conversion degree of 90% (i.e. \(c/c_0=0.1\)) of the lactose, when \(c_0=0.15\ \text{M}\). An amount of enzyme is added corresponding to \(v_{\max}=300\ \mu\text{M/min}\).
12. Compute how long the reaction time becomes for an inhibited process.
13. The found reaction time seems too long, since one will not accept a reaction time of more than four hours. How large must \(v_{\max}\) be if one wants to ensure that the process has proceeded within 4 hours?
4 Reaction conditions#
The rate of chemical reactions depends on temperature, which also applies to enzyme-catalyzed reactions. In addition, enzymes (as proteins) are sensitive to high temperatures, since they will denature (lose activity) when exposed to high temperatures (like egg white that coagulates when eggs are boiled). The quantity \(v_{\max}\) will now depend on reaction time and temperature.
The enzymes are added in the form of a liquid preparation with a specified strength, which depends on reaction temperature and decreases with reaction time. The relation between \(v_{\max}(t,T)\) and the enzyme strength \(E(t,T)\) is given by
where \(V_e, V_r\) indicate respectively the volume of added enzyme and the volume of the reaction mixture. \(T\) denotes the absolute temperature measured in Kelvin (K). In the following we use \(V_r=1000\ \text{L}\).
We introduce the initial strength
For the β-galactosidase preparation, for Celsius temperatures in the interval \([0^\circ\text{C},50^\circ\text{C}]\), it holds that
where \(T_1\) is an empirical constant. The time-dependent denaturation of the enzyme can be described by
where \(k(T)\) is a rate constant that depends on temperature, since
\(T_2\) is also an empirical constant. If the time- and temperature-dependent \(v_{\max}(t,T)\) is inserted into equation (4), one obtains
14. Set up an expression for \(c(t,T)\) and \(t(c,T)\) for the complete inhibited process given by (9).
15. Fix the Celsius temperature at \(35^\circ\text{C}\) and let \(c_0=0.15\ \text{M}\). An amount of enzyme corresponding to \(V_e=0.1\ \text{L}\) is added. Compute the concentration \(c\) as a function of reaction time, and draw the graph of the function. Explain what happens when \(t\to\infty\).
16. Fix again \(c_0=0.15\ \text{M}\). Investigate how long the reaction time must be to achieve a conversion degree of 90% at a Celsius temperature of respectively \(30^\circ\text{C}\), \(35^\circ\text{C}\) and \(40^\circ\text{C}\) when \(V_e=0.1\ \text{L}\). Compute how much enzyme must be added to reach a conversion degree of 90% within four hours.
5 Optimization#
The choice of reaction parameters for conversion of lactose to galactose and glucose is not free, since there are limitations on reaction time at different temperatures, see Table 1. The reason for the limitation of reaction time is the risk of growth of undesirable microorganisms. The longer reaction time at \(50^\circ\text{C}\) is due to the fact that very few troublesome microorganisms grow at such a high temperature.
Table 1: The maximum reaction time at different temperatures
Celsius temperature, \(^\circ\text{C}\) |
Maximum reaction time (hours) |
|---|---|
5 |
24 |
10 |
10 |
20–45 |
4 |
50 |
8 |
The final problems now concern determining sets of reaction conditions that minimize the costs. Reaction conditions here mean the amount of added enzyme, the reaction time and the temperature.
The cost \(O\) to carry out the process is initially assumed only to depend on the added enzyme amount. Explicitly, we assume
We want to optimize the costs under the following conditions:
The Celsius temperature must lie in the interval \([5^\circ\text{C},50^\circ\text{C}]\).
The reaction time must not exceed the conditions given in Table 1.
The initial concentration is \(c_0=0.15\ \text{M}\).
The conversion degree must be 90% (\(c/c_0=0.1\)).
These requirements imply that \(V_e\) can be written as a function of reaction time and temperature, i.e. \(V_e(t,T)\), where \((t,T)\) lies within a region in the plane that satisfies the conditions in Table 1.
17. Find the minimum and minimizer for the function \(O\) under the given conditions.
The total costs to carry out the process of course also depend on costs of operating the plant (including the plant price, maintenance, temperature control in the process, etc.). We assume that this additional cost for the individual process is proportional to the time it takes to complete the process. The total cost is therefore now
18. Set \(\beta=1\ \text{kr/minute}\). Find the minimum and minimizer for \(O\) under the same conditions as before.
